
Perbedaan Isotop, Isobar, Isoton, dan Isomer Muhyidin, SKM
Isotopes and Isobars. Isobar are elements that differ in chemical properties but have the same physical property. So, we can say that isobars are those elements that have a different atomic number but the same mass number. In contrast, Isotopes are those elements having the same atomic number and different mass numbers.

ISOTOP, ISOBAR, ISOTON, dan ISOELEKTRON STRUKTUR ATOM YouTube
v. t. e. Nuclide half-lives colorcoded. Two nuclides are isotones if they have the same neutron number N, but different proton number Z. For example, boron-12 and carbon-13 nuclei both contain 7 neutrons, and so are isotones. Similarly, 36 S, 37 Cl, 38 Ar, 39 K, and 40 Ca nuclei are all isotones of 20 because they all contain 20 neutrons.

Cara Menentukan Isotop Isobar Dan Isoton Studyhelp
Iodine isobars are used to treat goiter. Cobalt isobars can be used to treat cancer. Frequently Asked Questions. Q1. What are isotopes and isobars? Answer. Isotopes are atoms of the same element with the same atomic number but a different mass number. Isobars are atoms of various elements that have the same mass number but a different atomic.
Isotopes and Isobars Definition, Uses and Difference Teachoo
Isotop, Isobar, Isoton: Pengertian dan Contohnya. Berdasarkan nomor atom dan nomor massanya, atom-atom dari unsur yang sama ataupun berbeda dapat dibedakan menjadi isotop, isobar, dan isoton. Seperti diketahui, dalam ilmu kimia, kita diperkenalkan dengan konsep atom yaitu bagian terkecil dari suatu materi. Partikel dasar penyusun atom adalah.

Isotop, isobar,dan isoton kimia SMA YouTube
Perbedaan Isotop Isobar dan IsotonMateri terkait Struktur Atom1. Bilangan kuantum Utama, Azimut, Magnetik, dan Spinhttps://www.youtube.com/watch?v=ESqI8jDl57.
Isotop, Isobar and Isoton (Kimia SBMPTN, UN, SMA) YouTube
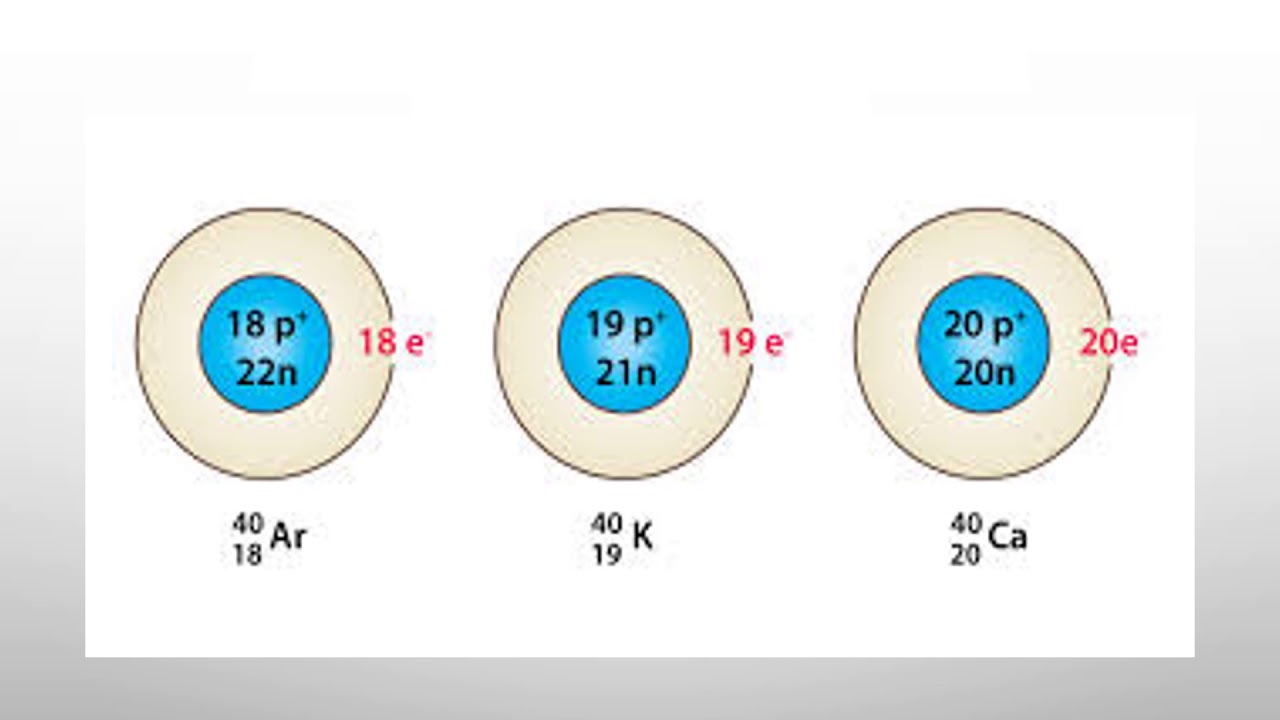
Isobars are elements that have the same number of nucleons (sum of protons and neutrons). The series of elements with 40 Mass numbers serve as a good example; 40 16 S, 40 17 Cl, 40 18 Ar, 40 19 K, and 40 20 Ca. The nucleus of all the above-mentioned elements contain the same number of particles in the nucleus but contain varying numbers of.

Contoh Isotop Isobar Dan Isoton cara mengatasi kaki pegal saat mau tidur
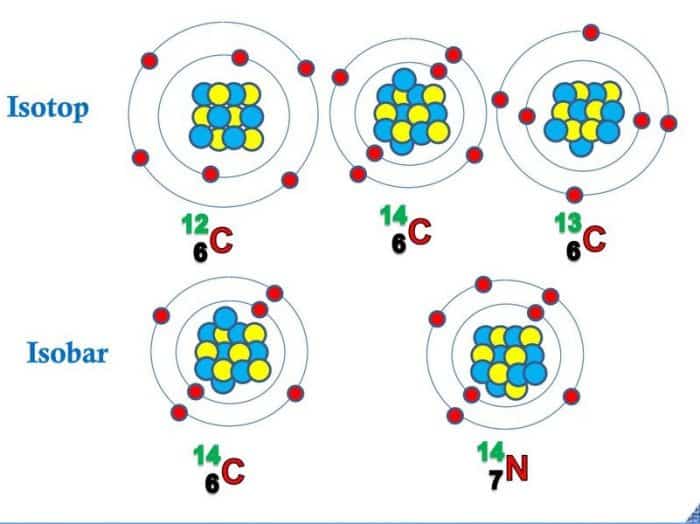
Pengertian Isobar: unsur atomnya berbeda namun memiliki nomor massa yang sama. Hal ini dinamakan isobar. Contoh Isobar. Natrium dan Magnesium dapat mempunyai nomor massa yang sama yaitu 24 Na 11 dan 24 Mg 12 Hidrogen dan Helium dapat mempunyai nomor massa yang sama yaitu 3 H 1 dan 3 He 2 Karbon dan Nitrogen dapat mempunyai nomor massa yang sama yaitu 14 C 6 dan 14 N 7. C. Isoton

Struktur Atom Kimia Kelas 10 • Part 3 Isotop Isobar Isoton, Massa Atom & Molekul Relatif YouTube
Isobars Elements having the same mass number (A) but different number of protons (Z) are isobars. Example: 40 16 S, 40 17 Cl, 40 18 Ar, 40 19 K, and 40 20 Ca. Isotones Elements having the same number of neutrons (N) but a different number of protons (Z) or mass number (A) are isotones. Example: 36 16 S, 37 17 Cl, 38 18 Ar, 39 19 K, and 40 20 Ca.

Isotop, Isobar, dan Isoton My Chemistry ff
ISOBARS Isobar is a group of nuclides that has sum of proton and neutron are same but sum of each proton and neutron is different. • Examples : 12C6 with 12 C 7 • Examples : 12C6 with 12 C 7 5.

Pembahasan Soal Isotop, Isobar, dan Isoton Struktur Atom YouTube
Isobars. The set of elements has the same number of nucleons, where nucleons are protons or neutrons. For example, 40 Sulfur, 40 Chlorine, 40 Argon, 40 Potassium, and 40 Calcium are all isobars. Moreover, despite having the same mass number, isobars have different atomic numbers for different chemical elements.
Pengertian Serta Contoh Isotop, Isobar dan Isoton Siswapedia
Thus, the major difference between Isotopes and Isobars is that isotopes belong to the same element while isotones are different elements. Also, isotopes have similar chemical properties. However, isobars have different chemical properties. For example, 12 C, 13 C, and 14 C are the isotopes of Carbon while 26 Fe 58 and 27 Ni 58 are Isotones.

Structure of Atom (Part 1) Isotopes, isotones and Isobar Class 11, chapter 2 YouTube
Apa perbedaan isotop, isobar dan isoton? Jawaban: Isotop. Dilansir dari Encyclopaedia Britannica, isotop adalah atom dengan nomor atom yang berbeda namun menduduki tempat yang sama pada table periodik. Jadi unsur pada table periodik yang kamu lihat sebeneranya tidak hanya satu unsur, melainkan ada dua atau lebih unsur yang duduk pada posisi.

Difference Between Isotopes And Isobars Understanding The Key Differences A Plus Topper
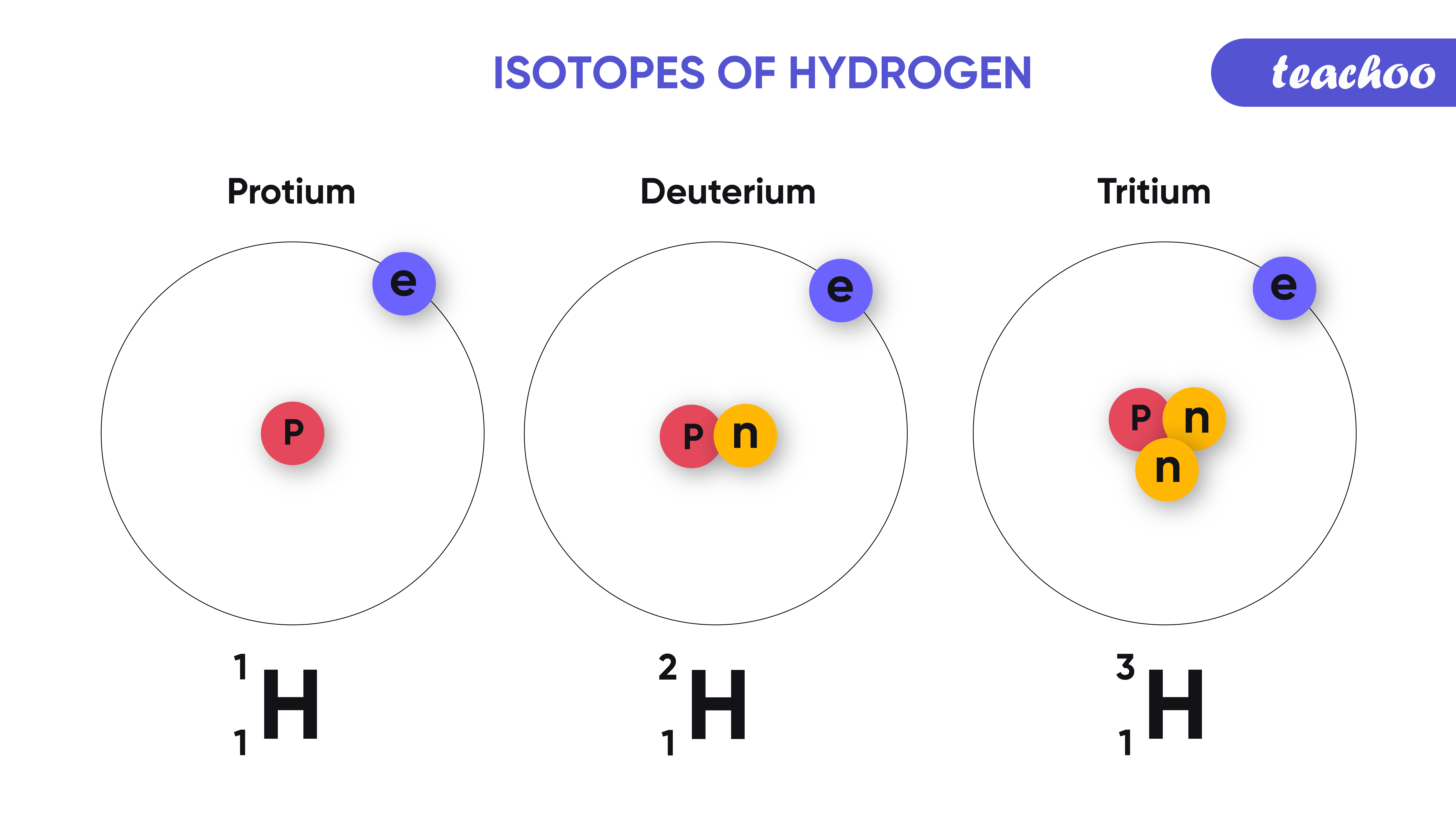
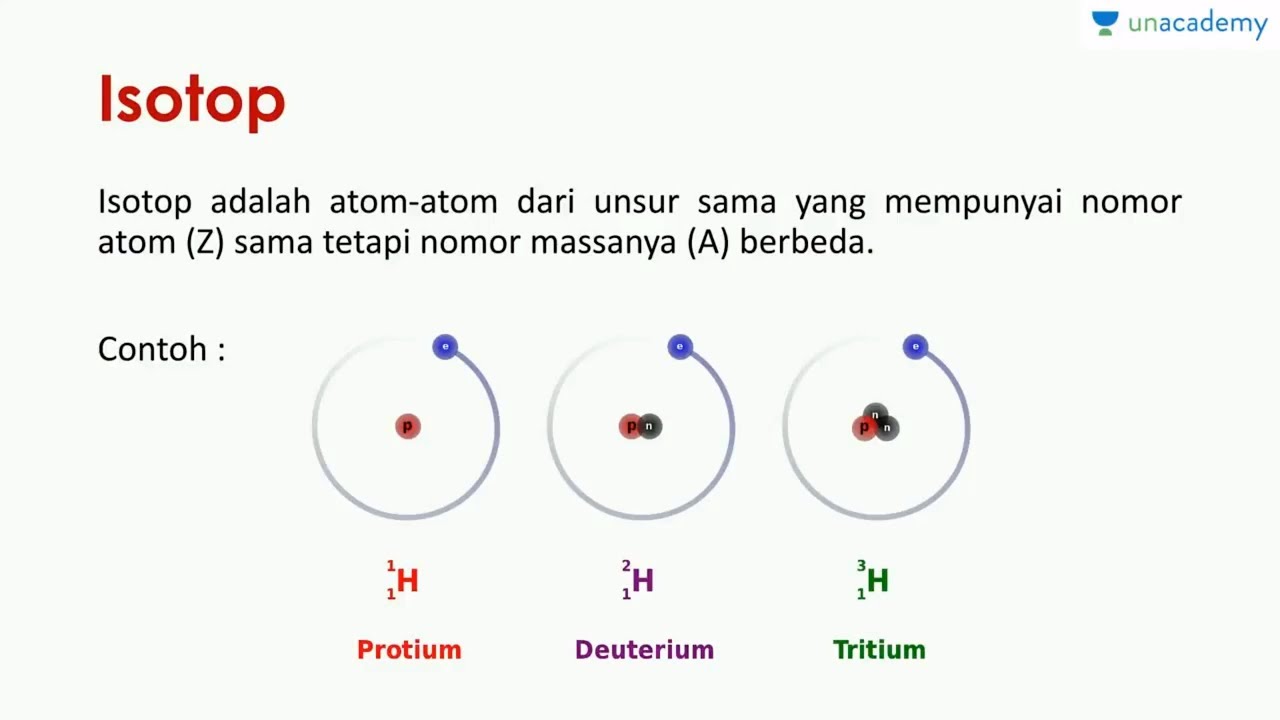
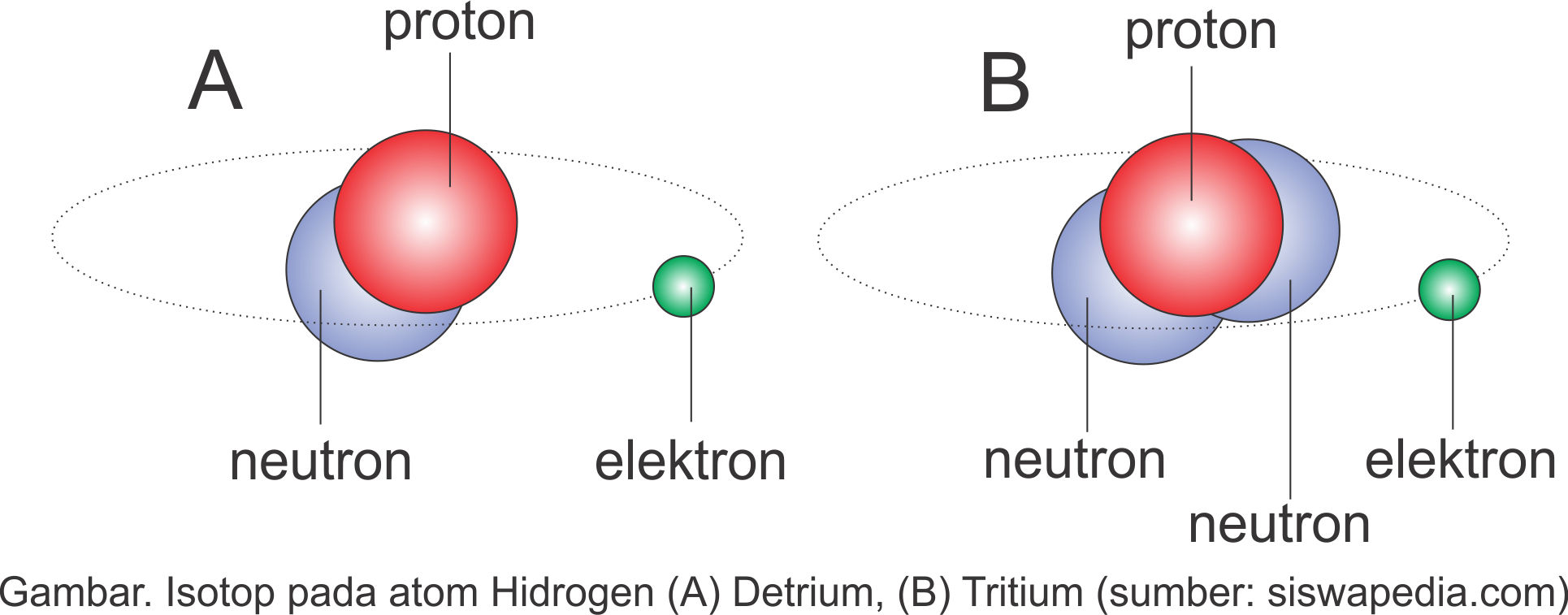
Isotopes. Isotopes are atoms of an element which have the same proton number but different nucleon numbers. Example: Hydrogen is the common example which has three isotopes. These have the same atomic number, one, but different mass numbers 1, 2, and 3. These three isotopes are commonly known as hydrogen or protium, deuterium (D) and tritium (T.
Isotope, Isobar and Isotone pptx YouTube
Memahami Definisi Isotop, Isobar dan Isoton dalam Ilmu Kimia Beserta Contoh. 27 Oktober 2023. Yusuf Abdhul Azis. Pada saat pertama kali mengenal istilah isotop, isobar, dan juga isoton ini mungkin kamu akan sedikit bingung bahkan pusing. Apalagi ada sedikit perbedaan antara isotop, isobar dan juga isoton. Meskipun begitu, dewasa ini proses.
Teori Struktur Atom Menurut Para Ahli Beserta Pengertiannya (Lengkap)
ISOTOPES, ISOBARS AND ISOTONES87 Thus Aston's Mass spectrograph not only helped in identifying the isotopes present in an element but also helped in determining the average atomic mass of a given element. 18 20 22 24 26 28 30 32 34 36 38 Neon Chlorine Mass spectrographs of Neon and Chlorine. Figure 3.2 SOLVED PROBLEM.

10 Differences between isotopes and isobars DewWool
As mentioned before, isotopes are atoms that have the same atomic number, but different mass numbers. Isotopes are denoted the same way as nuclides, but they are often symbolized only with the mass numbers because isotopes of the same element have the the same atomic number. Carbon, for example, has two naturally occurring isotopes, 12 6 C and.